Last year, when I attended ATTD 2021, I wrote about Dr. Walter Pories and his observations in people with type 2 diabetes undergoing gastric bypass surgery. You can read that article here but, in short, obese people with type 2 diabetes, immediately after gastric bypass surgery, begin seeing significant improvements in insulin resistance/requirements. Literally in the days following surgery insulin requirement drops by a factor of 10 and, within a month, remission often occurs. Now the surgery does not remove the fat, visceral or otherwise, and yet there is such a dramatic improvement in insulin needs.
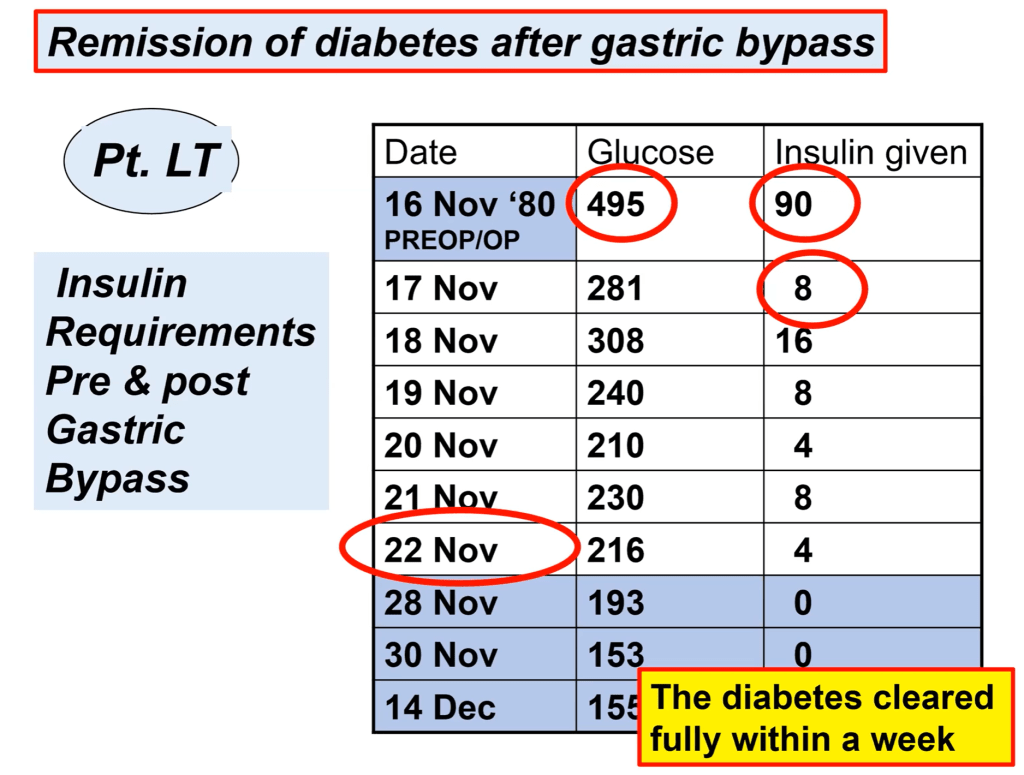
Dr. Pories concluded type 2 diabetes was not caused by “lifestyle” or visceral fat deposits but by a faulty gut which processed food poorly generating excess fat and little energy. The net result is increased weight, low energy levels, and overeating to compensate generating a vicious cycle. This subsequently strains the pancreas in certain individuals whose pancreas cannot keep up and we have type 2 diabetes.
Rather than being a disease of the lazy or a disease of the gluttonous, if Dr Pories is right, type 2 diabetes is a disease of the gut.
This month I was fortunate enough to attend ISPAD 2022 as a dedoc Voice and it turns out there is a controversy with type 1 diabetes which also challenges the cause.
Conventional Wisdom On The Cause Of Type 1 Diabetes.
The conventional thinking on the cause of type 1 diabetes begins with what we can observe and that is the immune system. Generally speaking, in people with type 1 diabetes, we see auto-antibody markers. In other words, the body’s immune system is attacking the pancreas and, specifically, the beta cells.
The idea often put forward is an “environmental factor” interacts with the body somehow, and the immune system responds to what it believes is a threat. As the immune system is a learning system, it remembers the threat and is prepared to attack it in the future. The theory is that the threat looks similar to the beta cells in the body and so the immune system begins attacking them, leading to depletion and type 1 diabetes.
That environmental factor might be a virus, or it could be gluten causing a “leaky gut”, exposing the immune system to food particles and confusing it. Professor Bart Roep is suggesting pinning it on the “environmental factor” is missing the mark.
What If The Beta Cells Were The Problem?
Professor Roep gave his talk in the first session of the first day and set quite the benchmark.

He is looking at how we are approaching cancer treatment and whether the lessons learned there can be used for the treatment of type 1 diabetes. Rather than seeing type 1 diabetes as caused by a confused immune system, he suggests it is caused by stressed beta cells.
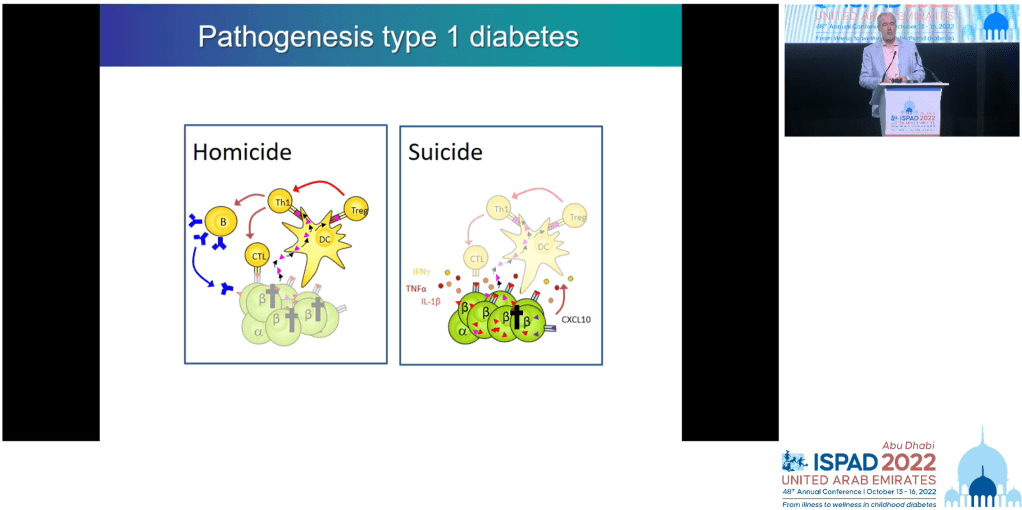
To explain this he cited the late Gian Franco Bottazzo who promoted a radical idea; rather than the immune system causing indiscriminate destruction of beta cells (homicide), the beta cells are literally asking to be killed (as also happens with cancer cells) and the immune system is obliging.
Rebalancing The Immune System
When I was first diagnosed with type 1 diabetes, and told it was a problem of the immune system, my first thought was to turn to immune suppression drugs, as we do with organ transplants to prevent the immune system from attacking the new organ. So why do we not use immune suppressions drugs to slow the destruction of the pancreas? Because, in this case, the treatment is considered worse than the disease.
There are significant side effects from immune suppression drugs not the least of which is an increased risk of cancer. It turns out the body’s cells know they are cancerous or infected by a virus and literally put their hand up to be killed by the immune system. When we suppress the immune system this goes unchecked; infections and cancers are left unhindered and compromise the host.
On the other side of the coin, if we boost the immune system, this can trigger a disease very similar to type 1 diabetes.
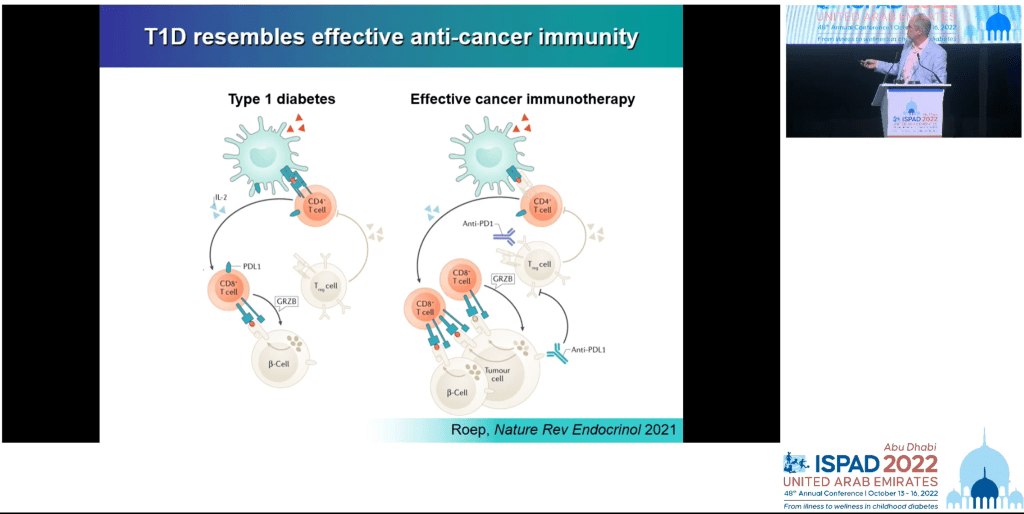
The idea being that the immune system becomes much more sensitive to stressed cells and attacks cells with less discrimination. Great for cancer but not so great for beta cells.
Beta Cells Are Stressed
So why are beta cells the outlier in being attacked? Why does the immune system focus here and not on the entire body? It turns out beta cells are some of the hardest working cells in the human body producing literally a million copies of insulin every minute.
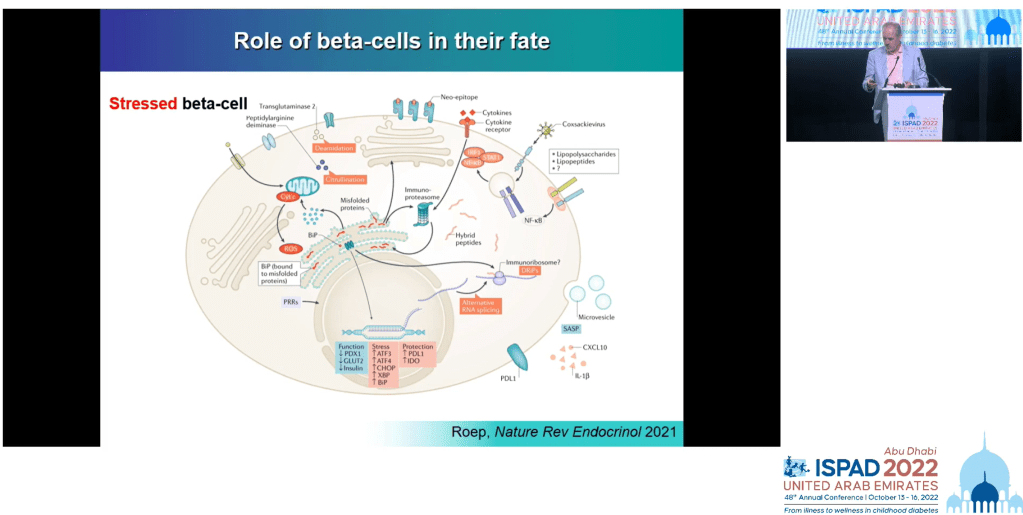
It therefore makes sense that it would not take a lot to tip a beta cell over the stress threshold and be targeted by the immune system.
Evidence That Stress Is The Problem
LADA forums frequently advise that to prolong the honeymoon (time when beta cells are still producing substantial amounts of insulin), you must reduce the stress on the beta cells. Often recommendations include dietary adjustments, such as eating low carbohydrate, supplements to reduce inflammation, and early-intervention insulin to reduce the requirement of the beta cells.
Even in the medical literature there is evidence (or, at least, consistency) that beta cell stress accelerates the destruction of beta cells. In “Latent Autoimmune Diabetes in Adults: A Review on Clinical Implications and Management”, a summary of findings from other studies included:
- Insulin sensitizers plus insulin therapy preserve beta cells function better than insulin alone
- Progression to insulin dependence was slower when insulin therapy was used compared to the use of sulfonylureas (a class of drugs which force beta cells to produce more insulin)
“Beta-Cell Preservation…Is Weight Loss the Answer?” presents evidence that weight loss can lower insulin resistance and preserve beta cell function.
All of the above findings are consistent with this stress model for Type 1 diabetes.
Similarly, this model does not exclude things like viruses as being involved, it simply recasts their role as stressors of the beta cells rather than antagonists of the immune system.
The Opportunity For New Treatments

Looking at cancer treatment breakthroughs and applying an inverse approach we get a novel set of approaches for curing/managing type 1 diabetes.
CAR T cells
In cancer, the immune cells (T cells) are reengineered to attack cancer cells. For type 1 diabetes, the reengineering would be with the cells that regulate the T cells (Regulatory T cells) to supress the immune response specifically for beta cell destruction.
Bionic: therapeutic Ab conjugated with toxin
In cancer, antibodies are engineered to carry a toxin so when they attack the cancer cell they deliver a toxic payload to wipe out the cancel cell. This same technique would be used but to deliver a growth factor to revitalise the beta cell.
Reduce hypo inflammatory tumor environment
Cancer cells actively reduce inflammation to limit the immune response. Treatments often seek to reverse this. In the case of type 1 diabetes, treatments would look to enhance the low inflammatory state.
DC vaccination (Dendreon)
In cancer, a company called Dendreon has a vaccine which selectively activates the immune system. For type 1 diabetes, we can selectively suppress the immune system through the use of vitamin D3.
In the case of this last approach Professor Roep’s team has tested it with excellent results; the general immune system was preserved but the immune component responsible for attacking the pancreas was suppressed.
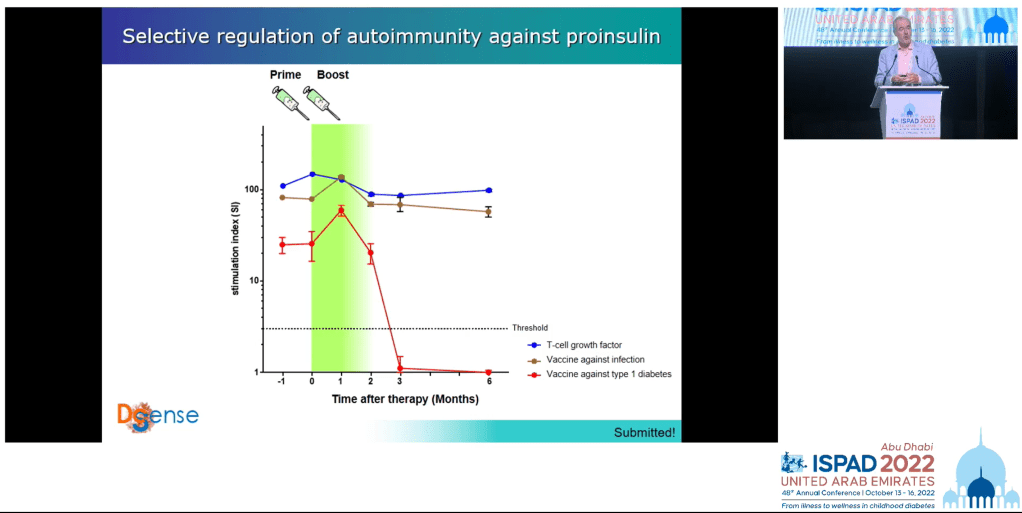
Professor Roep also made the point that having a variety of treatments means they can be tailored to individuals, just as cancer drugs provide a set of tools in the toolkit of the oncologist with patients responding to different ones.
Conclusions
While I enjoyed the entirety of ISPAD 2022, this was, for me, the stand out presentation. Challenging the type 1 origin story, presenting a model which is consistent with a broad range of observations, and pioneering resulting therapies is, for me, what these conferences are all about and I look forward to seeing the progress of Professor Roep’s work at conferences in the future.
Thank you so much for this amazing article summarising and explaining this session so understandably! I loved this session as it is really interesting to see different approaches and conclusions I never heard about before! B. Roep is an amazing speaker and it is a pleasure listening to him.
LikeLiked by 1 person
Reblogged this on Closed loop – Open mind! and commented:
Großartiger Artikel… Über eine tolle, kontroverse Session beim ISPAD2022
LikeLike
Don’t understand how D3 can suppress immune system… It is supposedly good for you…
I am taking about 8000 IU (which is 200 micro g of D3) weekly… Am I suppresing my immune system…
I live in Ireland where there is not a lot of sun, so I had deficency of D3. With this dose I keep my D3 at right level.
LikeLike
Hi Andy,
This is what was suggested in the talk. A bit of digging around confirms it is an immune modulator which can help with inflammation and immune disorders (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3194221/). Skip to the Concluding Remarks as there is a LOT of text there.
LikeLike