I am coming to the end of my EASD 2020 series but what a conference it was for announcements and discoveries. It just goes to show that research into diabetes is thriving and it suggests we have a lot to look forward to in the future with continual advancements in medical treatments and technology,
The presentations on Insulin Icodec, a once-weekly basal insulin, was particularly exciting for me.
What is Insulin Icodec?
Insulin has come a long way in the last 100 years. Early forms of insulin were derived from farm animals but, with the advance of recombinant DNA (the ability to combine DNA from multiple sources), it became possible to manufacture insulin with bacteria or yeast. Mass production of ‘regular’ insulin (human-equivalent) followed until the mid-nineties. In 1996 a new form of insulin was introduced to the market by Eli Lilly in the form of ‘Lispro rDNA’. This was a genetically modified form of insulin which acted in the body like regular insulin but with modified properties.
Since then, insulin analog advances have been made to improve how quickly a rapid-acting insulin takes to reach peak activity or, in the case of basal insulins, to make them last for longer and more consistently in the body.
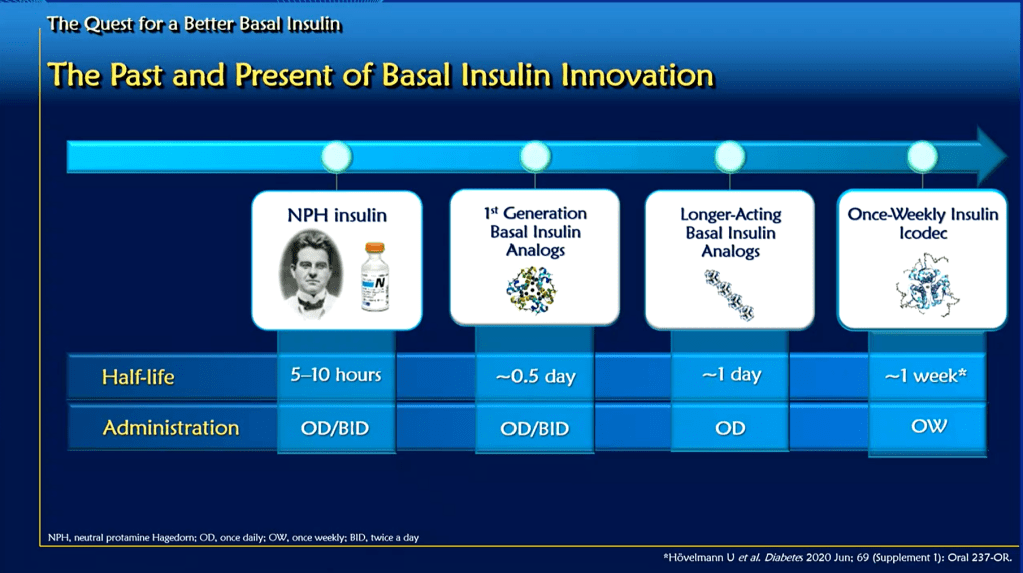
Insulin Icodec is very much in this second camp. While some basal insulins, up until now, could work for around 24 hours, this is not the case for Icodec which makes basal insulin available to the body over a period of one week.
Presentations
Ulrike Hoevelmann presented on the effectiveness of the insulin. As we can see here, it maintains a relatively constant supply of insulin to the body over the seven days.

This particular research involved 50 participants and explored the properties of Icodec compared to Degludec (Tresiba).
Julio Rosenstock presented on a study with 247 participants comparing daily basal injections with weekly injections in a double-blind, double-dummy test. Once-weekly insulin Icodec showed a higher rate of hypoglycaemic events, compared to daily basal insulin glargine U100.
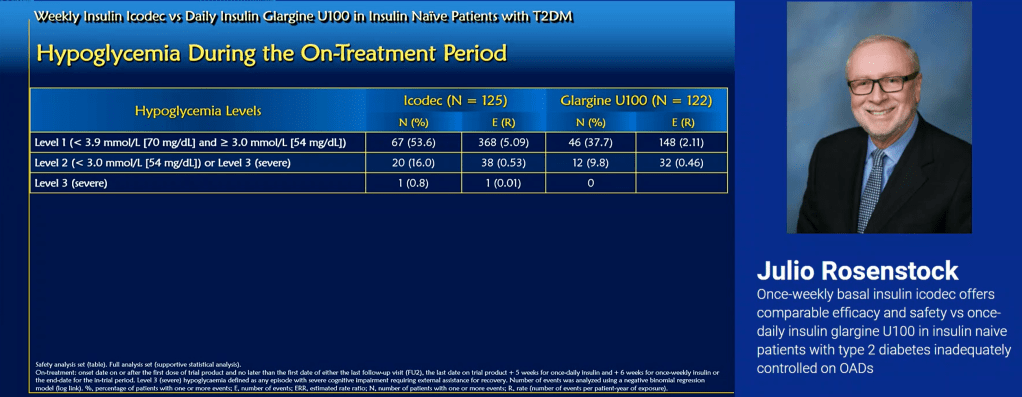
However, Doctor Rosenstock was quick to point out the only difference which was statistically significant was the Level 1 hypos. In all other aspects compared, there was no statistically significant difference in outcomes or adverse events between the two insulins.

Only For Type 2s?
Both studies focussed exclusively on Type 2s. So, will Icodec also work for other Types, such as Type 1? I asked this in one of the presentations and was told it certainly will but this is not where they are researching right now. My guess is the research trials are focussing on the majority of diabetics i.e. Type 2s. It is still early days so I am sure, before Icodec reaches the market, it will also be tested in Type 1s. While some Type 1s I know have expressed concerns around the risks of being subject to a bad dosing decision for an entire week, I think this would affect all patients about the same, regardless of Type. Personally, I think the more technology can remove the burden of management from diabetics and carers, the better and going from 365 injections a year down to 52, for me, is compelling.
This is quite encouraging! I just wonder why there haven’t been advancements in oral insulin (where it’s protected from stomach acid until it hits the intestines).
If diabetes were a disease treated simply by taking a pill a few times/day, then it wouldn’t be considered any more of an epidemic than high cholesterol or hypertension, right?
LikeLike
It’s a good question. There is now an oral GLP-1 Agonist (https://practicaldiabetic.com/2020/10/10/the-benefits-of-incretin-mimetics-for-all-diabetics/) which changes the local pH in the gut to protect the medication so perhaps an oral insulin is the next step.
LikeLike